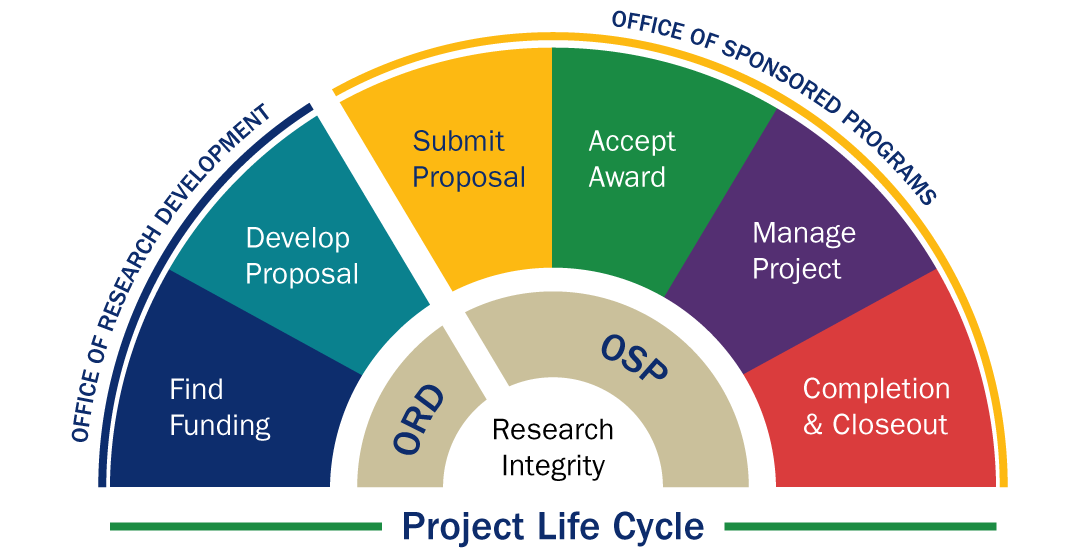
Office of Sponsored Programs (OSP)
Welcome to OSP -- we are here to help!
Learn more about OSP >
Office of Research Development (ORD)
The Office of Research Development (ORD) aims to catalyze innovative and impactful scholarship by increasing competitiveness for sponsored research, enhancing research capacities, and facilitating cultures of collaboration, inclusivity, and creativity.
Learn more about ORD >
Research Integrity & Compliance (RIC)
Research Integrity & Compliance (RIC) oversees University programs to ensure compliance with federal, state, and local regulations for research. RIC creates and supports an environment that furthers the ethical and responsible conduct of research.
Learn more about RIC >
Upcoming Events:









