Catalysis
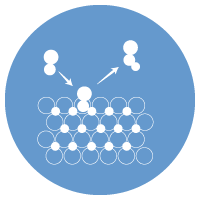

Catalysts increase rate and reduce energy required for chemical conversion. Researchers in the Energy Research Institute are developing and modeling catalysts to perform tasks such as transforming small molecules into value added compounds including polymers and synthesizing platform and specialty chemicals from waste lignocellulosic biomass. Catalytic systems being developed include transition metal catalysts (including methods to prevent coarsening of nanoparticles at higher temperatures) and zeolite templated materials.
Researchers

James Crawford is an Assistant Professor in the Chemical and Biological Engineering Department at MSU. He is interested in the catalytic transformation of gaseous molecules (CO₂, CO, CH₄, H₂O, H₂, N₂, O₂) into high value fuels, chemicals, and building materials. He is especially interested in studying the similarity between metal-zeolite catalysts and metalloenzymes. Through bioinspired synthesis, in operando characterization, and reaction testing our group hopes to unlock new catalytic possibilities.
Dr. Crawford can be reached at 406-994-7378 or [email protected].
Website: https://crawford-lab.com/

Sharon Neufeldt is an assistant professor in the Chemistry and Biochemistry Department. Research in the Neufeldt group targets the design, optimization, and understanding of catalytic systems for transforming small molecules into value-added compounds such as sequence-controlled polymers and fuels. In particular, her group is interested in homogeneous transition metal catalysts for achieving highly selective or unusual reactions. Research areas include catalyst-controlled chemoselectivity in cross-coupling reactions and developing catalysts that can functionalize methane by new mechanistic pathways. In addition to standard organic, organometallic, and inorganic chemistry lab techniques, we also use computational tools to gain insight into the structure and reactivity of catalytic systems.
Dr. Neufeldt can be reached at 406-994-5692 or [email protected].
Website: http://www.chemistry.montana.edu/people/Neufeldt-Sharon.html

Cecily Ryan is an assistant professor in the Mechanical and Industrial Engineering Department. Her research interests are: biodegradable and bio-derived polymers and composites; materials properties of composites, including interfacial compatibilization; additive manufacturing of biobased composites; biointerfaces with polymeric materials; sustainable material and energy product lifecycles; incorporation/reuse of waste materials; environment-material interaction; multi-functional materials; and nanocomposites.
Dr. Ryan can be reached at 406-994-7272 or [email protected].

Dr. Stephen Sofie is assistant professor in the Mechanical & Industrial Engineering Department. Metal electro-catalysts represent the standard for high performance, low cost high temperature fuel cell electrodes. Moving towards solution infiltrated catalysts to achieve nano-scale (10-80 nm), high surface area electro-catalyst coverage, yields some detriment to the use of nano-metallic catalysts. Thermodynamic degradation and hence coarsening of fine catalyst particles can lead to catalyst attrition and performance drops, ultimately limiting the long term stability of these nano-scale materials at temperatures up to 900C. Research activities are examining novel approaches to stabilizing nano-metal catalysts by the incorporation of tailored secondary phases at the catalyst/support interface to mitigate degradation by physically binding the catalyst. Mixtures of aluminum and titanium oxides have been shown to react in-situ to fuel cell electrolyte materials forming complex functional oxides that dramatically enhance fuel cell electrode longevity.
Dr. Sofie can be reached at 406-599-4481 or [email protected].

Nicholas Stadie is an assistant professor in the Chemistry and Biochemistry Department. The Stadie group explores new synthesis routes toward structurally and chemically tunable carbon-based materials with a chief interest in their application for energy storage. The group’s interests are in both electrochemical energy storage (e.g., porous carbon-based batteries) and gas storage (e.g., in adsorbed phases of hydrogen or methane under high pressures), as well as gas separations and catalysis. Contributing fundamental insights into gas adsorption phenomena at the solid interface is also a goal of the group’s work, using simple thermodynamics and statistical mechanics models.
Dr. Stadie can be reached at 406-994-7931 or [email protected].
Website: http://www.montana.edu/stadiegroup/

Dr. Stephanie Wettstein is an assistant professor in Chemical & Biological Engineering Department. The overall goal of Dr. Wettstein's research is to increase the sustainability of biofuels and chemicals produced from lignocellulosic biomass by improving processing methods, increasing reaction rates, and increasing the yields of biomass carbon that is converted into biofuels and chemicals. This includes developing novel, high yield biomass deconstruction methods, improved biomass conversion processes to increase catalyst stability, and separation methods using zeolite membranes.
Dr. Wettstein can be reached at 406-994-5928 or [email protected].
Website: http://www.chbe.montana.edu/staff/wettstein/index.html